Bioinformatics for cancer research

Understand cancer, develop new drugs and personalize treatment with Genevia Technologies.
Our history in cancer bioinformatics is long: we have had the privilege to work with biologists seeking for a deeper understanding of cancer, companies developing new treatments to cancer, as well as oncologists wishing to optimize therapies to individual patients.
Below you will find highlights of our journey in cancer research. If you feel inspired and would like to benefit from our expertise, leave us a message and we will book you a call with our bioinformatician.
Leave us a short description of your bioinformatics needs and we will be in touch very soon!
Understanding cancer
What causes cancer? Which alterations in DNA, pathways and metabolic processes allow a tumor to grow, spread and evade treatment? How does tumorigenic reprogramming relate to normal cellular differentiation?
Our experience in cancer biology covers research into fundamental questions across cancer types and high-throughput molecular data types. Together with our collaborators and customers, we have studied heritable and somatic variants and their downstream molecular effects as well as the evolution and microenvironment of tumors, to name but a few aspects of cancer biology.
Whether you are setting out to characterize an understudied malignancy or dive deep into the molecular biology of a more common cancer, we have you covered, bioinformatically speaking.
Learn more
- RNA-seq data analysis
- Single-cell RNA-seq data analysis
- DNA-seq data analysisMutation analysis
- Clonality analysis
- Epigenomic data analysis
- Proteomic and metabolomic data analysis
References and case studies
Selected publications from our customers
- Hunold, P. et al. (2025). DynaTag for efficient mapping of transcription factors in low-input samples and at single-cell resolution. Nature communications, 16(1), 6585. https://doi.org/10.1038/s41467-025-61797-9
- Mallikarjuna, P. et al. (2025). Liquid biomarkers associate with TGF-β Type I receptor and hypoxia in kidney cancer. Signal transduction and targeted therapy, 10(1), 309. https://doi.org/10.1038/s41392-025-02404-7
- Chang, Y. T. et al. (2024). MHC-I upregulation safeguards neoplastic T cells in the skin against NK cell-mediated eradication in mycosis fungoides. Nature communications, 15(1), 752. https://doi.org/10.1038/s41467-024-45083-8
- Häyrinen, M. et al. (2024). Tumor RNA sequencing identifies a group of Mycosis Fungoides patients with failure of skin-directed therapies. The Journal of investigative dermatology, S0022-202X(24)02084-0. Advance online publication. https://doi.org/10.1016/j.jid.2024.06.1292
- Boyd, S., et al. (2024). NGS of brush cytology samples improves the detection of high-grade dysplasia and cholangiocarcinoma in patients with primary sclerosing cholangitis: A retrospective and prospective study. Hepatology communications, 8(4), e0415. https://doi.org/10.1097/HC9.0000000000000415
- Chérouvrier Hansson, V. et al. (2024). Dichotomous Effects of Glypican-4 on Cancer Progression and Its Crosstalk with Oncogenes. International journal of molecular sciences, 25(7), 3945. https://doi.org/10.3390/ijms25073945
- Häyrinen, M. J. et al. (2023). The Transcription Factor Twist1 Has a Significant Role in Mycosis Fungoides (MF) Cell Biology: An RNA Sequencing Study of 40 MF Cases. Cancers, 15(5), 1527. https://doi.org/10.3390/cancers15051527
- Karihtala, P. et al. (2022). Comparison of the mutational profiles of neuroendocrine breast tumours, invasive ductal carcinomas and pancreatic neuroendocrine carcinomas. Oncogenesis, 11(1), 53. https://doi.org/10.1038/s41389-022-00427-1
- Song, J. et al. (2022). The ubiquitin-ligase TRAF6 and TGFβ type I receptor form a complex with Aurora kinase B contributing to mitotic progression and cytokinesis in cancer cells. EBioMedicine, 82, 104155. https://doi.org/10.1016/j.ebiom.2022.104155
- Yuan, O. et al. (2022). A somatic mutation in moesin drives progression into acute myeloid leukemia. Science advances, 8(16), eabm9987. https://doi.org/10.1126/sciadv.abm9987
- Wahlström, G. et al. (2022). The variant rs77559646 associated with aggressive prostate cancer disrupts ANO7 mRNA splicing and protein expression. Human molecular genetics, ddac012. Advance online publication. https://doi.org/10.1093/hmg/ddac012
- Tusup, M. et al. (2022). Epitranscriptomics modifier pentostatin indirectly triggers Toll-like receptor 3 and can enhance immune infiltration in tumors. Molecular therapy : the journal of the American Society of Gene Therapy, 30(3), 1163–1170. https://doi.org/10.1016/j.ymthe.2021.09.022
- Kundu, S. et al. (2021). Common and mutation specific phenotypes of KRAS and BRAF mutations in colorectal cancer cells revealed by integrative -omics analysis. Journal of experimental & clinical cancer research : CR, 40(1), 225. https://doi.org/10.1186/s13046-021-02025-2
- Tikkanen, T. et al. (2018). Seshat: A Web service for accurate annotation, validation, and analysis of TP53 variants generated by conventional and next-generation sequencing. Human mutation, 39(7), 925–933. https://doi.org/10.1002/humu.23543
Selected publications from our team
Vlachavas, E. I. Et al. (2025). Molecular and functional profiling unravels targetable vulnerabilities in colorectal cancer. Molecular oncology, 10.1002/1878-0261.13814. Advance online publication. https://doi.org/10.1002/1878-0261.13814
Brunner, E. et al. (2025). Unraveling the YAP1-TGFβ1 axis: a key driver of androgen receptor loss in prostate cancer-associated fibroblasts. bioRxiv 2025.02.25.640167; doi: https://doi.org/10.1101/2025.02.25.640167
Schwarzmüller, L et al. (2025). PAK1 activation drives divergent resistance mechanisms to aromatase inhibition and Tamoxifen in a luminal A breast cancer model. bioRxiv 2025.04.28.651009; doi: https://doi.org/10.1101/2025.04.28.651009
Kiviaho, A. et al. (2024). Single cell and spatial transcriptomics highlight the interaction of club-like cells with immunosuppressive myeloid cells in prostate cancer. Nature communications, 15(1), 9949. https://doi.org/10.1038/s41467-024-54364-1
Marteau, V. et al. (2024). Single-cell integration and multi-modal profiling reveals phenotypes and spatial organization of neutrophils in colorectal cancer. bioRxiv 2024.08.26.609563; doi: https://doi.org/10.1101/2024.08.26.609563
Fotakis, G. et al. (2024). Conventional therapy induces tumor immunoediting and modulates the immune contexture in colorectal cancer. bioRxiv 2024.08.21.608938; doi: https://doi.org/10.1101/2024.08.21.608938
Obacz, J. et al. (2024). IRE1 endoribonuclease signaling promotes myeloid cell infiltration in glioblastoma. Neuro-oncology, 26(5), 858–871. https://doi.org/10.1093/neuonc/noad256
Salcher, S. et al. (2024). Comparative analysis of 10X Chromium vs. BD Rhapsody whole transcriptome single-cell sequencing technologies in complex human tissues. Heliyon, 10(7), e28358. https://doi.org/10.1016/j.heliyon.2024.e28358
Nurminen, A. et al. (2023). Cancer origin tracing and timing in two high-risk prostate cancers using multisample whole genome analysis: prospects for personalized medicine. Genome medicine, 15(1), 82. https://doi.org/10.1186/s13073-023-01242-y
Plattner, C. et al. (2023). Functional and spatial proteomics profiling reveals intra- and intercellular signaling crosstalk in colorectal cancer. iScience, 26(12), 108399. https://doi.org/10.1016/j.isci.2023.108399
Caronni, N. et al. (2023). IL-1β+ macrophages fuel pathogenic inflammation in pancreatic cancer. Nature, 623(7986), 415–422. https://doi.org/10.1038/s41586-023-06685-2
Beumers, L. et al. (2023). Clonal heterogeneity in ER+ breast cancer reveals the proteasome and PKC as potential therapeutic targets. NPJ breast cancer, 9(1), 97. https://doi.org/10.1038/s41523-023-00604-4
Ricordel, C. et al. (2023). Genomic characteristics and clinical significance of CD56+ circulating tumor cells in small cell lung cancer. Scientific reports, 13(1), 3626. https://doi.org/10.1038/s41598-023-30536-9
Simigdala, N. et al. (2023). Loss of Kmt2c in vivo leads to EMT, mitochondrial dysfunction and improved response to lapatinib in breast cancer. Cellular and molecular life sciences : CMLS, 80(4), 100. https://doi.org/10.1007/s00018-023-04734-7
Aakula, A. et al. (2023). RAS and PP2A activities converge on epigenetic gene regulation. Life science alliance, 6(5), e202301928. https://doi.org/10.26508/lsa.202301928
Dundr, P. et al. (2023). Uterine leiomyoma with RAD51B::NUDT3 fusion: a report of 2 cases. Virchows Archiv : an international journal of pathology, 10.1007/s00428-023-03603-9. Advance online publication. https://doi.org/10.1007/s00428-023-03603-9
Kontogianni, G. et al. (2023). A Comprehensive Analysis of Cutaneous Melanoma Patients in Greece Based on Multi-Omic Data. Cancers, 15(3), 815. https://doi.org/10.3390/cancers15030815
Rodriguez-Martinez, A. et al. (2022). Novel ZNF414 activity characterized by integrative analysis of ChIP-exo, ATAC-seq and RNA-seq data. Biochimica et biophysica acta. Gene regulatory mechanisms, 1865(3), 194811. Advance online publication. https://doi.org/10.1016/j.bbagrm.2022.194811
Salcher, S. et al. (2022). High-resolution single-cell atlas reveals diversity and plasticity of tissue-resident neutrophils in non-small cell lung cancer. Cancer cell, 40(12), 1503–1520.e8. https://doi.org/10.1016/j.ccell.2022.10.008
Kukkonen, K. et al. (2022). Nonmalignant AR-positive prostate epithelial cells and cancer cells respond differently to androgen. Endocrine-related cancer, 29(12), 717–733. https://doi.org/10.1530/ERC-22-0108
Machado-Lopez, A. et al. (2022). Integrative Genomic and Transcriptomic Profiling Reveals a Differential Molecular Signature in Uterine Leiomyoma versus Leiomyosarcoma. International journal of molecular sciences, 23(4), 2190. https://doi.org/10.3390/ijms23042190
Gao, Y. et al. (2022). Inactivation of AMPK Leads to Attenuation of Antigen Presentation and Immune Evasion in Lung Adenocarcinoma. Clinical cancer research : an official journal of the American Association for Cancer Research, 28(1), 227–237. https://doi.org/10.1158/1078-0432.CCR-21-2049
Andersson, A. et al. (2021). Spatial deconvolution of HER2-positive breast cancer delineates tumor-associated cell type interactions. Nature communications, 12(1), 6012. https://doi.org/10.1038/s41467-021-26271-2
Hall, Z. et al. (2021). Lipid Remodeling in Hepatocyte Proliferation and Hepatocellular Carcinoma. Hepatology (Baltimore, Md.), 73(3), 1028–1044. https://doi.org/10.1002/hep.31391
- Taavitsainen, S. et al. (2021). Single-cell ATAC and RNA sequencing reveal pre-existing and persistent cells associated with prostate cancer relapse. Nature communications, 12(1), 5307. https://doi.org/10.1038/s41467-021-25624-1
- Filppu, P. et al. (2021). CD109-GP130 interaction drives glioblastoma stem cell plasticity and chemoresistance through STAT3 activity. JCI insight, 6(9), e141486. https://doi.org/10.1172/jci.insight.141486
- Vlachavas, E. I. et al. (2021). A Detailed Catalogue of Multi-Omics Methodologies for Identification of Putative Biomarkers and Causal Molecular Networks in Translational Cancer Research. International journal of molecular sciences, 22(6), 2822. https://doi.org/10.3390/ijms22062822
Woodcock, D. J. et al. (2020). Prostate cancer evolution from multilineage primary to single lineage metastases with implications for liquid biopsy. Nature communications, 11(1), 5070. https://doi.org/10.1038/s41467-020-18843-5
- Dufva, O. et al. (2020). Immunogenomic Landscape of Hematological Malignancies. Cancer cell, 38(3), 380–399.e13. https://doi.org/10.1016/j.ccell.2020.06.002
- Mehtonen, J. et al. (2020). Single cell characterization of B-lymphoid differentiation and leukemic cell states during chemotherapy in ETV6-RUNX1-positive pediatric leukemia identifies drug-targetable transcription factor activities. Genome medicine, 12(1), 99. https://doi.org/10.1186/s13073-020-00799-2
- Yoosuf, N. et al. (2020). Identification and transfer of spatial transcriptomics signatures for cancer diagnosis. Breast cancer research : BCR, 22(1), 6. https://doi.org/10.1186/s13058-019-1242-9
Fotakis, G. et al. (2020). NeoFuse: predicting fusion neoantigens from RNA sequencing data. Bioinformatics (Oxford, England), 36(7), 2260–2261. https://doi.org/10.1093/bioinformatics/btz879
- Pölönen, P. et al. (2019). Hemap: An Interactive Online Resource for Characterizing Molecular Phenotypes across Hematologic Malignancies. Cancer research, 79(10), 2466–2479. https://doi.org/10.1158/0008-5472.CAN-18-2970
- Vlachavas, E. I. et al. (2019). Radiogenomic Analysis of F-18-Fluorodeoxyglucose Positron Emission Tomography and Gene Expression Data Elucidates the Epidemiological Complexity of Colorectal Cancer Landscape. Computational and structural biotechnology journal, 17, 177–185. https://doi.org/10.1016/j.csbj.2019.01.007
- Escobar, G. et al. (2018). Interferon gene therapy reprograms the leukemia microenvironment inducing protective immunity to multiple tumor antigens. Nature communications, 9(1), 2896. https://doi.org/10.1038/s41467-018-05315-0
- Norelli, M. et al. (2018). Monocyte-derived IL-1 and IL-6 are differentially required for cytokine-release syndrome and neurotoxicity due to CAR T cells. Nature medicine, 24(6), 739–748. https://doi.org/10.1038/s41591-018-0036-4
- Berglund, E. et al. (2018). Spatial maps of prostate cancer transcriptomes reveal an unexplored landscape of heterogeneity. Nature communications, 9(1), 2419. https://doi.org/10.1038/s41467-018-04724-5
- de Bock, C. E. et al. (2018). HOXA9 Cooperates with Activated JAK/STAT Signaling to Drive Leukemia Development. Cancer discovery, 8(5), 616–631. https://doi.org/10.1158/2159-8290.CD-17-0583
- Gao, Q. et al. (2018). Driver Fusions and Their Implications in the Development and Treatment of Human Cancers. Cell reports, 23(1), 227–238.e3. https://doi.org/10.1016/j.celrep.2018.03.050
- Määttä, K. et al. (2016). Whole-exome sequencing of Finnish hereditary breast cancer families. European journal of human genetics : EJHG, 25(1), 85–93. https://doi.org/10.1038/ejhg.2016.141





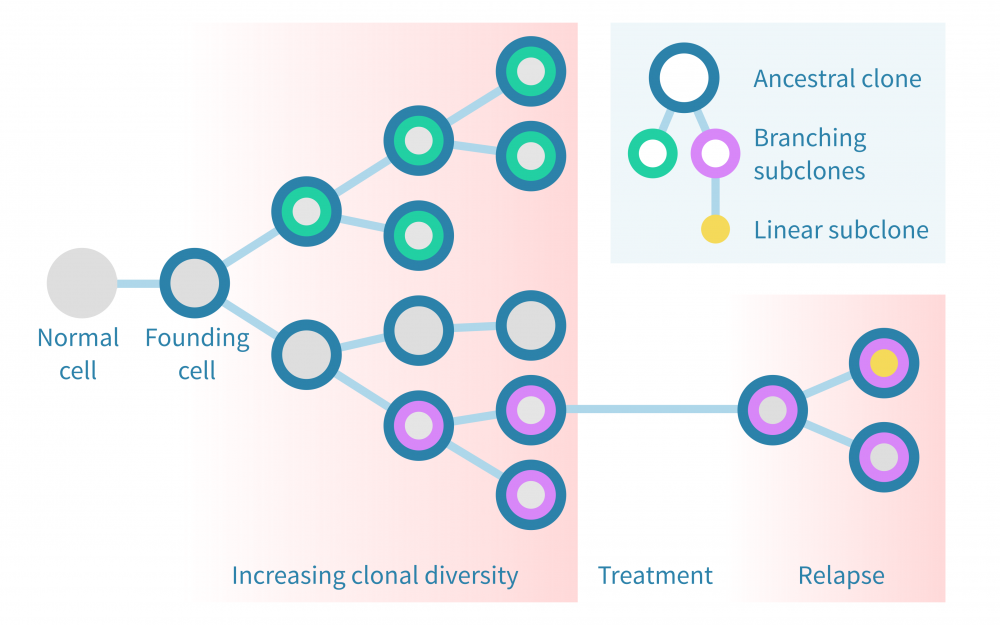
Treating cancer
Developing a new cancer therapy is a long, costly and risky process. High-throughput measurements coupled with cutting-edge bioinformatics has a lot to offer along the way to both speed up the process and to increase the chances of success.
We can help in identifying targets for a given disease based on public and proprietary molecular and clinical data. Public data on molecular drug perturbation profiles, on the other hand, enables scanning for new applications for pharmaceuticals that are already on the market.
For both preclinical and clinical research on a new treatment, transcriptomic, epigenomic and proteomic measurements can be used to study the molecular mechanism of action. This allows for further optimizing the treatment and ruling out off-target effects.
Learn more
- Bioinformatics for drug development
- Discovering targets and biomarkers for immunotherapies
- RNA-seq data analysis
- Single-cell RNA-seq data analysis
- Epigenomic data analysis
References and case studies
- Characterizing a Cancer Immunotherapy Target with Genomic Data (CDR-Life)
- Mechanism of action study (MinaTx)
- Transcriptome-based target identification (Barron Biomedical)
Selected publications from our team
Eigentler, A. et al. (2024). Glucocorticoid treatment influences prostate cancer cell growth and the tumor microenvironment via altered glucocorticoid receptor signaling in prostate fibroblasts. Oncogene, 43(4), 235–247. https://doi.org/10.1038/s41388-023-02901-5
Heidegger, I. et al. (2022). Comprehensive characterization of the prostate tumor microenvironment identifies CXCR4/CXCL12 crosstalk as a novel antiangiogenic therapeutic target in prostate cancer. Molecular cancer, 21(1), 132. https://doi.org/10.1186/s12943-022-01597-7
- Annala, M. et al. (2021). Cabazitaxel versus abiraterone or enzalutamide in poor prognosis metastatic castration-resistant prostate cancer: a multicentre, randomised, open-label, phase II trial. Annals of oncology : official journal of the European Society for Medical Oncology, 32(7), 896–905. https://doi.org/10.1016/j.annonc.2021.03.205
Fotakis, G. et al. (2020). NeoFuse: predicting fusion neoantigens from RNA sequencing data. Bioinformatics (Oxford, England), 36(7), 2260–2261. https://doi.org/10.1093/bioinformatics/btz879
Fotakis, G. et al. (2021). Computational cancer neoantigen prediction: current status and recent advances. Immuno-oncology technology, 12, 100052. https://doi.org/10.1016/j.iotech.2021.100052


Predicting outcomes
Being able to predict the onset and development of cancer enables better treatment through early and accurate diagnosis and personalized treatment.
We use survival analyses and machine learning approaches with clinical and molecular data to predict patient-specific risks. Such analyses result in biomarkers or multi-marker signatures with clinical potential.
Biomarker discovery projects have been some of the most fruitful and clinically promising ones we have participated in, as you may see from the references and publications below!
Learn more
- Discovering targets and biomarkers for immunotherapies
- Predicting cancer survival with machine learning
- Predicting treatment response with machine learning
References and case studies
- Cancer risk prediction with machine learning (Karolinska Institute)
- Cell therapy biomarker discovery (Kuopio Center for Gene and Cell Therapy)
- Prognostic methylation anaysis of uveal melanoma (Oslo University Hospital)
- Multiplex immunofluorescence analysis (Uppsala University)
- Extracellular vesicle-based biomarker discovery (FastEV)
- Investigating the role of glypicans in cancer
Selected publications from our customers
- Song, J. et al. (2025). Identification of progression markers for prostate cancer. Cell cycle (Georgetown, Tex.), 24(17-20), 382–399. https://doi.org/10.1080/15384101.2025.2563930
- Karihtala, P. et al. (2024). Mutational signatures and their association with cancer survival and gene expression in multiple cancer types. International journal of cancer, 10.1002/ijc.35148. Advance online publication. https://doi.org/10.1002/ijc.35148
- Nissinen, L. et al. (2024). Clustering of RNA co-expression network identifies novel long non-coding RNA biomarkers in squamous cell carcinoma. Scientific reports, 14(1), 16864. https://doi.org/10.1038/s41598-024-67808-x
- Karihtala, P. et al. (2023). Mutational signatures and their association with survival and gene expression in urological carcinomas. Neoplasia (New York, N.Y.), 44, 100933. Advance online publication. https://doi.org/10.1016/j.neo.2023.100933
- Cheng, F. et al. (2023). Attenuation of cancer proliferation by suppression of glypican-1 and its pleiotropic effects in neoplastic behavior. Oncotarget, 14, 219–235. https://doi.org/10.18632/oncotarget.28388
- Mezheyeuski, A. et al. (2023). An immune score reflecting pro- and anti-tumoural balance of tumour microenvironment has major prognostic impact and predicts immunotherapy response in solid cancers. EBioMedicine, 88, 104452. Advance online publication. https://doi.org/10.1016/j.ebiom.2023.104452
- Karihtala, P. et al. (2022). Mutational Signatures Associate With Survival in Gastrointestinal Carcinomas. Cancer genomics & proteomics, 19(5), 556–569. https://doi.org/10.21873/cgp.20340
- Pommergaard, H. C et al. (2022). Aldehyde dehydrogenase expression may be a prognostic biomarker and associated with liver cirrhosis in patients resected for hepatocellular carcinoma. Surgical oncology, 40, 101677. https://doi.org/10.1016/j.suronc.2021.101677
- Tsakonas, G. et al. (2021). High Density of NRF2 Expression in Malignant Cells Is Associated with Increased Risk of CNS Metastasis in Early-Stage NSCLC. Cancers, 13(13), 3151. https://doi.org/10.3390/cancers13133151
- Madonna, G. et al. (2021). Clinical Categorization Algorithm (CLICAL) and Machine Learning Approach (SRF-CLICAL) to Predict Clinical Benefit to Immunotherapy in Metastatic Melanoma Patients: Real-World Evidence from the Istituto Nazionale Tumori IRCCS Fondazione Pascale, Napoli, Italy. Cancers, 13(16), 4164. https://doi.org/10.3390/cancers13164164
Ness, C. et al. (2021). Integrated differential DNA methylation and gene expression of formalin-fixed paraffin-embedded uveal melanoma specimens identifies genes associated with early metastasis and poor prognosis. Experimental eye research, 203, 108426. https://doi.org/10.1016/j.exer.2020.108426
- Pommergaard, H. C. et al. (2021). Peroxisome proliferator-activated receptor activity correlates with poor survival in patients resected for hepatocellular carcinoma. Journal of hepato-biliary-pancreatic sciences, 28(4), 327–335. https://doi.org/10.1002/jhbp.745
- Lehto, T. K. et al. (2021). Transcript analysis of commercial prostate cancer risk stratification panels in hard-to-predict grade group 2-4 prostate cancers. The Prostate, 81(7), 368–376. https://doi.org/10.1002/pros.24108
Simao, F. et al. (2021). SRF-CLICAL: an approach for patient risk stratification using random forest models. bioRxiv 2021.06.22.448514; doi: https://doi.org/10.1101/2021.06.22.448514
Selected publications from our team
- Fonseca, N. M. et al. (2024). Prediction of plasma ctDNA fraction and prognostic implications of liquid biopsy in advanced prostate cancer. Nature communications, 15(1), 1828. https://doi.org/10.1038/s41467-024-45475-w
- Kallio, H. M. et al. (2024). Sensitive circulating tumor DNA-based residual disease detection in epithelial ovarian cancer. Life science alliance, 7(6), e202402658. https://doi.org/10.26508/lsa.202402658
- Cao, S., Wang et al. (2022). Estimation of tumor cell total mRNA expression in 15 cancer types predicts disease progression. Nature biotechnology, 10.1038/s41587-022-01342-x. Advance online publication. https://doi.org/10.1038/s41587-022-01342-x
- Rautajoki, K. J. et al. (2022). PTPRD and CNTNAP2 as markers of tumor aggressiveness in oligodendrogliomas. Scientific reports, 12(1), 14083. https://doi.org/10.1038/s41598-022-14977-2
- Vandekerkhove, G. et al. (2021). Plasma ctDNA is a tumor tissue surrogate and enables clinical-genomic stratification of metastatic bladder cancer. Nature communications, 12(1), 184. https://doi.org/10.1038/s41467-020-20493-6
- Taavitsainen, S. et al. (2019). Evaluation of Commercial Circulating Tumor DNA Test in Metastatic Prostate Cancer. JCO precision oncology, 3, PO.19.00014. https://doi.org/10.1200/PO.19.00014
- Kaikkonen, E. et al. (2018). ANO7 is associated with aggressive prostate cancer. International journal of cancer, 143(10), 2479–2487. https://doi.org/10.1002/ijc.31746

Meet some of our experts in cancer research
I am a Professor of Bioinformatics at Tampere University, where I lead the Computational Biology research group. My research focuses on developing algorithms and computational models to study the molecular basis of human cancers and to enable new diagnostics and treatments.
We leverage diverse omics data, statistical modeling, and machine learning to uncover the biological processes and mechanisms underlying disease. I work closely with experimental groups to validate and refine our hypotheses.
In the past two decades, my collaborative research endeavors have led to the several discoveries of genomic drivers and evolutionary patterns of prostate cancer.
I am a senior bioinformatics scientist with over 10 years of experience in immuno-oncology and tumor evolution, specializing in single-cell data analysis and scientific software engineering. My work centers on integrating and analyzing multimodal data, developing analysis workflows, creating scientific software, and modeling tumor evolution.
I have deep expertise in bulk and single-cell sequencing (WES/WGS, RNA-seq, ctDNA) and 3+ years of experience with spatial omics (transcriptomic and imaging-based). During my Ph.D. and postdoctoral research, I led multiple single-cell studies that resulted in high-impact publications and contributed to two cancer atlases (NSCLC and CRC).
I’m a senior computational biologist with 10+ years of experience in cancer bioinformatics and precision oncology, specializing in multi-omics data analysis (WES, RNA-seq, microarrays, proteomics) for biomarker discovery and translational research.
My work focuses on functional knowledge discovery—such as molecular footprinting to infer regulatory activity—and reconstructing tumor histories using public tools and clinical databases to support personalized medicine. I’m also skilled in statistics, R programming, and developing open-source, reproducible reports.
I am a bioinformatician specialised in cancer genomics and genetics with a 10-year experience analysing omics data in countless genetic, genomic, transcriptomic and epigenetic studies.
While my research focus has been in cancer, I have also gained experience in a number of other fields, such as immunology, aging and developmental biology. In recent years, I have also applied machine learning methods to harness biomedical data in various clinical applications.
I am a bioinformatics scientist specializing in oncology and human health data analysis, with over five years of experience in handling various omics data types. This includes bulk data (whole-exome, RNA-seq) and single-cell and spatial data (scRNA-seq, spatial transcriptomics, single-cell proteomics), as well as expertise in the experimental techniques to generate these types of data.
Throughout my career, I have successfully applied omics analysis to cancer and aging-related diseases, securing public grants, publishing in journals, and presenting at international conferences. I am highly motivated to solve complex challenges and advance healthcare research.
I am a senior bioinformatics scientist with over 10 years of experience in analyzing a wide range of next-generation sequencing (NGS) data types, including spatial transcriptomics, single-cell RNA-seq, bulk RNA-seq, ChIP-seq, CUT&Tag, ATAC-seq, single-cell ATAC-seq, MeDIP-seq, and BS-seq.
With a background in both mathematics and biology, I am well-equipped to analyze and interpret complex biological datasets. My work spans various fields, with significant contributions in immunology and oncology research.
Learn more
To continue exploring our background and expertise in cancer bioinformatics, here are a few suggestions:
Contact us
Leave your email address here with a brief description of your needs, and we will contact you to get things moving forward!







