Bioinformatics for microbiology
Explore microbial functions, communities and interactions.
Next-generation sequencing enables increasingly accurate identification of microbial identities, functions, and interactions.
We leverage the latest computational methods and databases to help microbiologists answer difficult questions. How does the genomic composition of microbes affect their functions and cross-species interactions? How can that information be used to discover new treatments or industrial applications?
Scroll down to learn how we help our customers with microbial bioinformatics. If you would like to benefit from our bioinformatics support, leave us a message!
Leave us a short description of your bioinformatics needs and we will be in touch very soon!
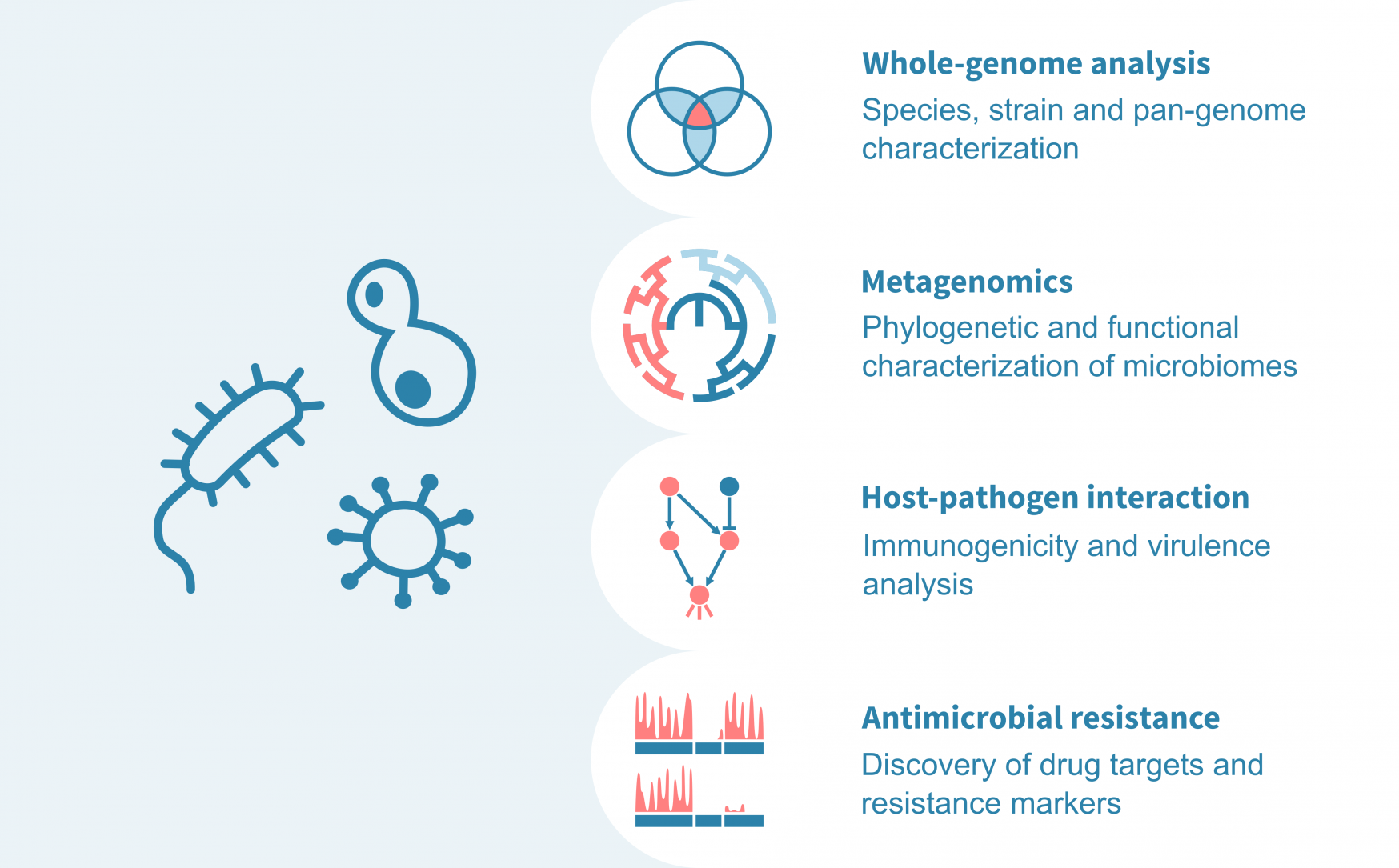
What do microbiologists use our bioinformatics service for?
An essential starting point for sequence-based microbial analyses lies in establishing a high-quality genome assembly, complete with functional annotations. This enables
- identifying species and strains,
- characterizing the shared and unique genes (core- and pan-genomes) of diverse strains,
- associating genomic and functional compositions to growth conditions or clinical variables, and
- identifying novel genes, particularly enzymes, for industrial or clinical applications.
For host-associated or environmental microbiomes, we analyze metagenomic sequence data to
- identify the microbes in the community (species / OTUs),
- characterize the phylogenetic and functional composition of the community, and
- compare the the diversities, functions and individual OTU abundances across conditions.
RNA-sequencing from isolated strains or microbiomes (metatranscriptomics) enables interrogating subtler changes in microbial functions even when the genomic composition remains the same.
For pathogenic microbes, we
- study the host's reaction to infection (from e.g., host RNA-sequencing data),
- identify virulence factors,
- identify genes essential for drug resistance (using e.g., transposon insertion sequencing data), and
- identify molecular markers (e.g., surface peptides) of drug resistance.
Learn more
References and case studies
- Investigating vector-borne diseases and pathogen transmission biology with transcriptomics (University of Florida)
- Bacterial genomics in bovine mastitits (University of Helsinki)
- Pharmacogenomic sequencing analysis (Karolinska Institute)
- Novel microbial enzyme discovery (Roal Oy)
- Virus quasispecies quantification (TATAA Biocenter)
- Consulting for cyanobacterial NGS data analysis (University of Turku)
Selected publications from our customers
- Ribeiro E Ribeiro, R. et al. (2024). Synchronous Epidermodysplasia Verruciformis and Intraepithelial Lesion of the Vulva Is Caused by Coinfection With Alpha-Human Papillomavirus and Beta-Human Papillomavirus Genotypes and Facilitated by Mutations in Cell-Mediated Immunity Genes. Archives of pathology & laboratory medicine, 148(9), 1014–1021. https://doi.org/10.5858/arpa.2023-0193-OA
- Åvall-Jääskeläinen, S. et al. (2021). Genomic Analysis of Staphylococcus aureus Isolates Associated With Peracute Non-gangrenous or Gangrenous Mastitis and Comparison With Other Mastitis-Associated Staphylococcus aureus Isolates. Frontiers in microbiology, 12, 688819. https://doi.org/10.3389/fmicb.2021.688819
- Pernaute-Lau, L. et al. (2021). Pharmacogene Sequencing of a Gabonese Population with Severe Plasmodium falciparum Malaria Reveals Multiple Novel Variants with Putative Relevance for Antimalarial Treatment. Antimicrobial agents and chemotherapy, 65(7), e0027521. https://doi.org/10.1128/AAC.00275-21
- Gallegos, J. E. et al. (2020). Challenges and opportunities for strain verification by whole-genome sequencing. Scientific reports, 10(1), 5873. https://doi.org/10.1038/s41598-020-62364-6
Selected publications from our team
- Kron, N. S. et al. (2024). Expression dynamics of the aplysia abyssovirus. Virology, 589, 109890. https://doi.org/10.1016/j.virol.2023.109890
- Saralahti, A. K. et al. (2023). Characterization of the innate immune response to Streptococcus pneumoniae infection in zebrafish. PLoS genetics, 19(1), e1010586. Advance online publication. https://doi.org/10.1371/journal.pgen.1010586
- L B Almeida, B. et al. (2022). The transcription factor network of E. coli steers global responses to shifts in RNAP concentration. Nucleic acids research, gkac540. Advance online publication. https://doi.org/10.1093/nar/gkac540
- Lindfors, K. et al. (2020). Metagenomics of the faecal virome indicate a cumulative effect of enterovirus and gluten amount on the risk of coeliac disease autoimmunity in genetically at risk children: the TEDDY study. Gut, 69(8), 1416–1422. https://doi.org/10.1136/gutjnl-2019-319809
- Harjula, S. E. et al. (2020). Characterization of immune response against Mycobacterium marinum infection in the main hematopoietic organ of adult zebrafish (Danio rerio). Developmental and comparative immunology, 103, 103523. https://doi.org/10.1016/j.dci.2019.103523
- Lin, J. et al. (2018). Bioinformatics Assembling and Assessment of Novel Coxsackievirus B1 Genome. Methods in molecular biology (Clifton, N.J.), 1838, 261–272. https://doi.org/10.1007/978-1-4939-8682-8_18
- Harjula, S. E. et al. (2018). Interleukin 10 mutant zebrafish have an enhanced interferon gamma response and improved survival against a Mycobacterium marinum infection. Scientific reports, 8(1), 10360. https://doi.org/10.1038/s41598-018-28511-w
- Havunen, R. et al. (2018). Abscopal Effect in Non-injected Tumors Achieved with Cytokine-Armed Oncolytic Adenovirus. Molecular therapy oncolytics, 11, 109–121. https://doi.org/10.1016/j.omto.2018.10.005
- Lin, J. et al. (2017). Vipie: web pipeline for parallel characterization of viral populations from multiple NGS samples. BMC genomics, 18(1), 378. https://doi.org/10.1186/s12864-017-3721-7
- Sin, C. et al. (2016). Quantitative assessment of ribosome drop-off in E. coli. Nucleic acids research, 44(6), 2528–2537. https://doi.org/10.1093/nar/gkw137
Genevia helped us to bring RNA-seq experiments in our R&D pipeline. By contracting this work, we were able to better focus our internal resources and still learn about sequencing and bioinformatics. We found that the Genevia team flexibly adapted to our requirements during the project.
We were in a need of a bioinformatics collaborator to advance the analysis of our cyanobacterial NGS data. Genevia Technologies provided us with flexible consulting on bioinformatics issues arising in our project. I appreciated the manner in which the project was executed, and would recommend Genevia to other academics.
The collaboration with Genevia proceeded well; they stayed on schedule and provided a well-structured project report. They sure have a wide range of expertise in their compact team. Genevia's team is always ready to react to the client's needs and help with the project design.
In our experience, outsourcing was efficient, and the results were provided fast. The deadlines were met beautifully, the quality of the work was spotless, and we had zero hiccups with our communication. So what is there not to recommend? We would choose Genevia Technologies again in a heartbeat.
Contact us
Leave your email address here with a brief description of your needs, and we will contact you to get things moving forward!

